When we hear the word “corrosion,” most people think about rust on iron. But in real engineering work, corrosion is much more complex. There are many types of corrosion, and each one happens due to a different chemical or physical reason.
Corrosion is an electrochemical process. This means it involves a reaction between metal, oxygen, moisture, and sometimes salts or chemicals. If we understand these corrosion types and mechanisms, we can control them and avoid failures in pipelines, tanks, bridges, and machines.
In this guide, you will learn the most common types of corrosion, how they happen, how to identify them, and how to prevent them.
Types of Corrosion and Their Mechanisms Explained
1. Uniform Corrosion (General Corrosion Type)
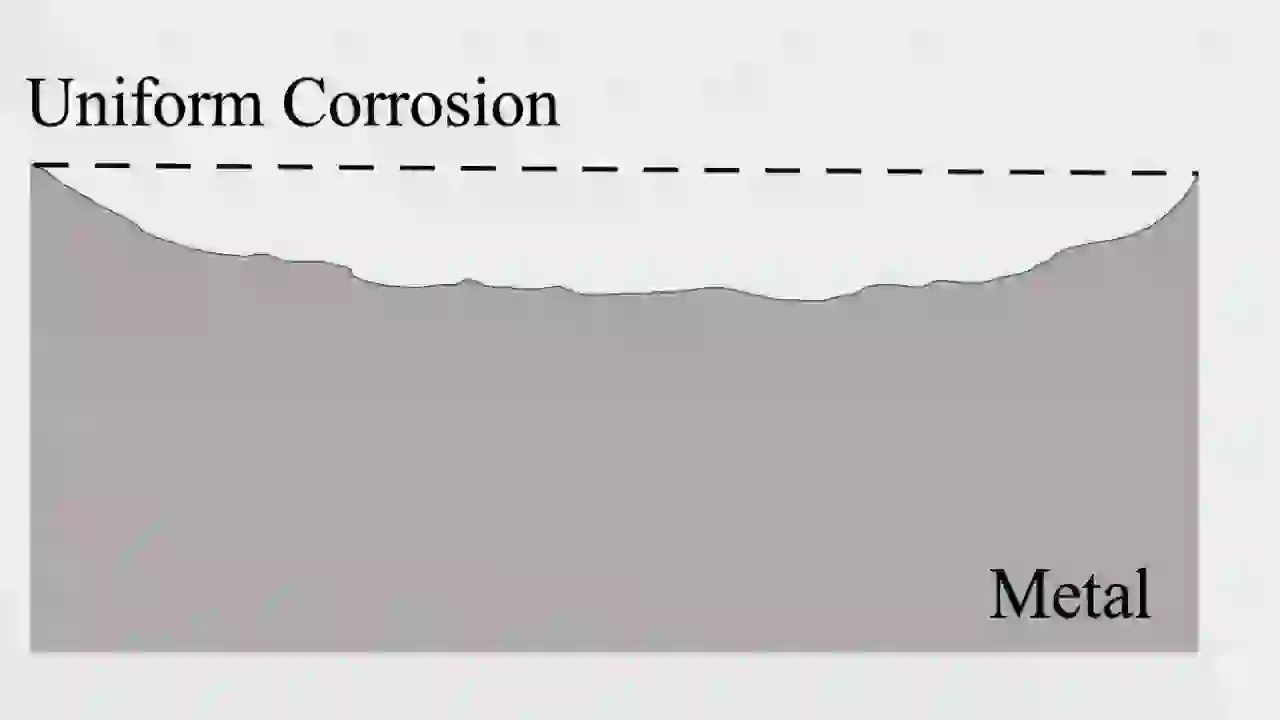
Uniform corrosion is the most common among all types of corrosion. In this case, the metal surface reacts evenly with the environment.
Technically, this happens because the anodic and cathodic reactions occur uniformly across the surface. The metal slowly loses thickness over time.
The look is simple—rust or material loss spread evenly across the surface. Engineers like this type because it is predictable. They can calculate corrosion rate using mm/year and plan maintenance.
Prevention mainly includes coatings, paints, and corrosion-resistant materials like stainless steel. Cathodic protection is also used in pipelines.
2. Galvanic Corrosion (Electrochemical Corrosion Type)
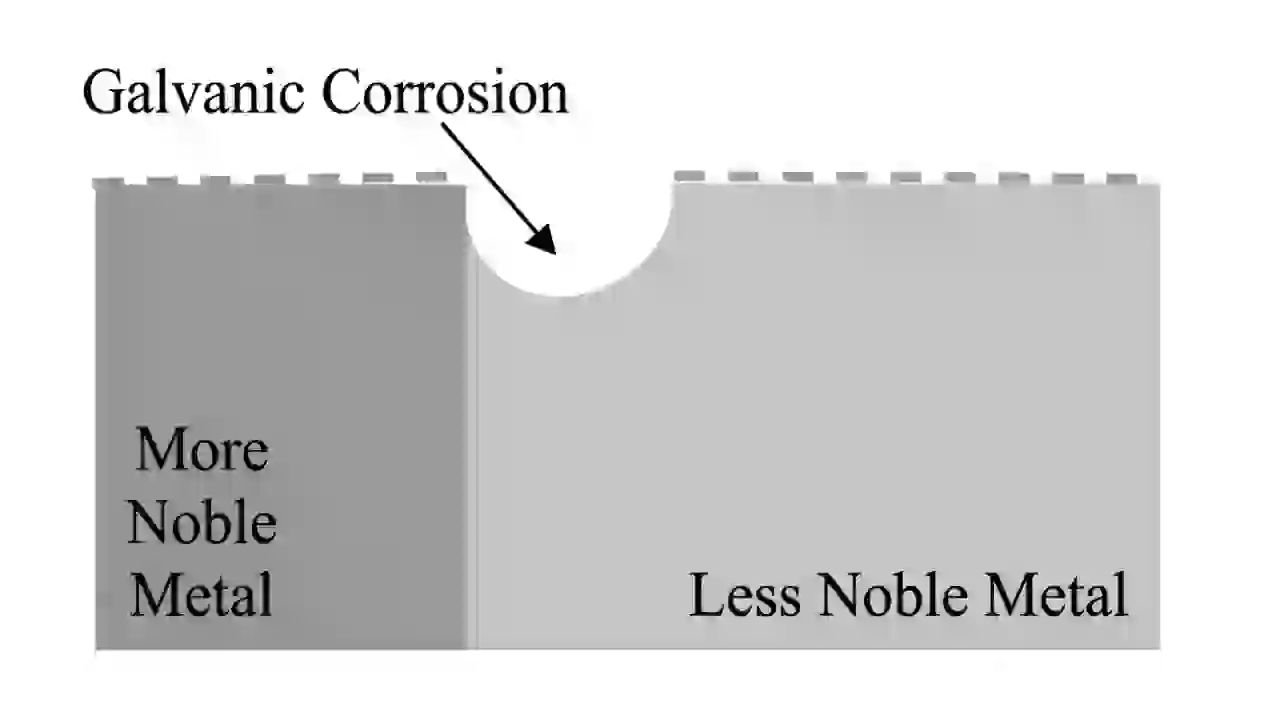
Galvanic corrosion happens when two different metals are connected in an electrolyte like water or saltwater. One metal becomes the anode and corrodes faster, while the other becomes the cathode and is protected.
This happens due to the difference in electrode potential between metals, which you can see in a galvanic series chart.
You will usually see corrosion near joints, bolts, or connections between dissimilar metals.
To prevent this corrosion type, engineers use insulation, select metals close in galvanic series, or apply coatings. Sacrificial anodes are also used in ships and pipelines.
3. Pitting Corrosion (Localized Corrosion Type)
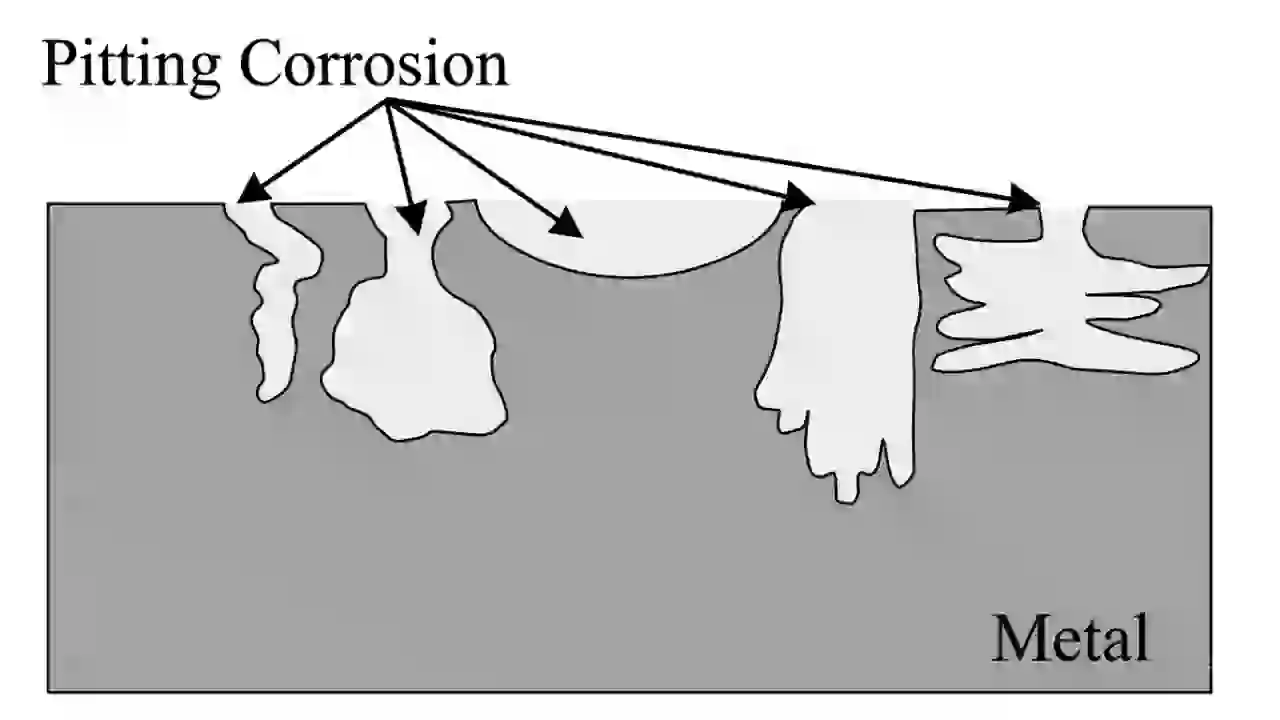
Pitting corrosion is one of the most dangerous corrosion types because it is very hard to detect.
It starts when the protective oxide layer on metal breaks locally. This creates a small anodic area, and corrosion goes deep into the metal.
Chloride ions (like in seawater) are the main reason for this corrosion mechanism, especially in stainless steel.
You will see very small holes on the surface, but inside the damage can be very deep. That is why it can cause sudden failure.
Prevention includes using pitting-resistant alloys, proper cleaning, and controlling chloride exposure.
4. Crevice Corrosion (Localized Corrosion in Gaps)
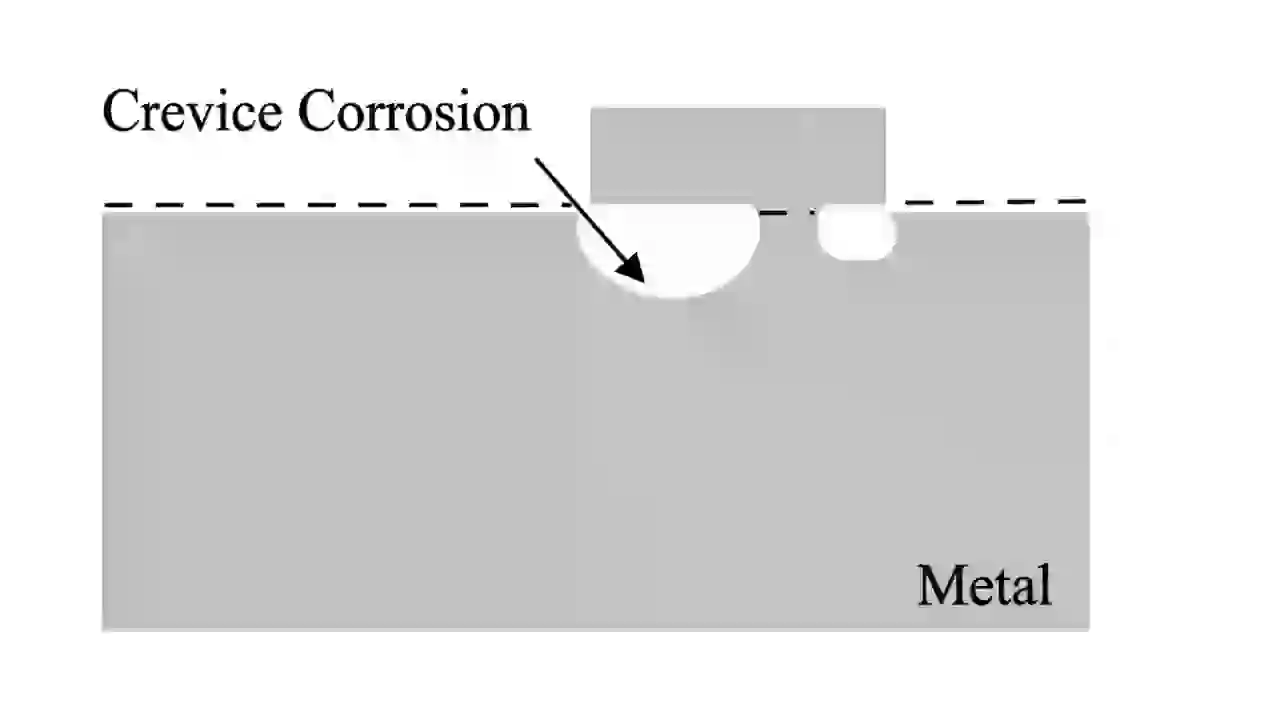
Crevice corrosion is another localized type of corrosion that occurs in small gaps where oxygen cannot enter easily.
Inside the crevice, oxygen level becomes low. This creates a difference in concentration, forming an electrochemical cell. The inside becomes anodic and starts corroding.
This is common under gaskets, washers, and lap joints.
The damage is usually hidden, which makes it dangerous. Prevention involves better design, sealing crevices, and using welded joints instead of bolted ones.
5. Intergranular Corrosion (Grain Boundary Corrosion Type)
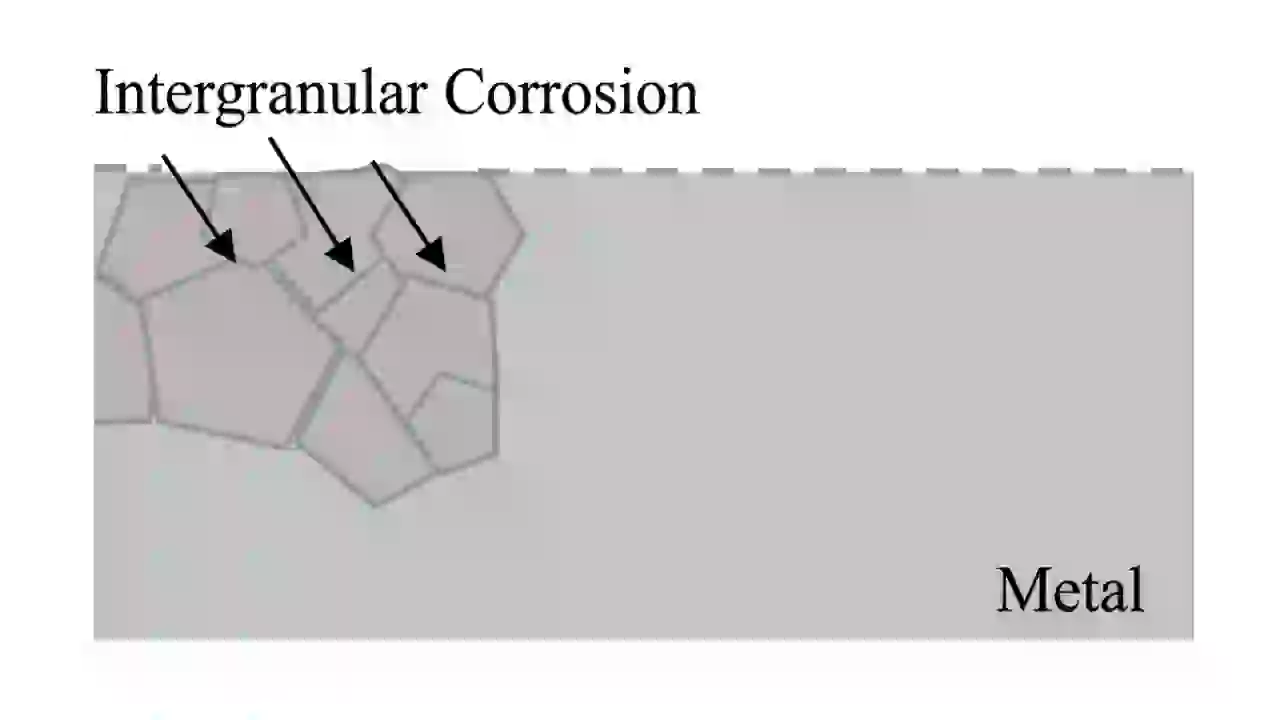
Intergranular corrosion affects the grain boundaries of metals, especially stainless steel.
This usually happens after welding when chromium carbides form at grain boundaries. This process is called sensitization. It reduces chromium in nearby areas, making them weak against corrosion.
The metal may look normal from outside but can break easily.
To prevent this corrosion type, low-carbon stainless steel (like 304L, 316L) is used. Proper heat treatment like solution annealing also helps.
6. Filiform Corrosion (Underfilm Corrosion Type)
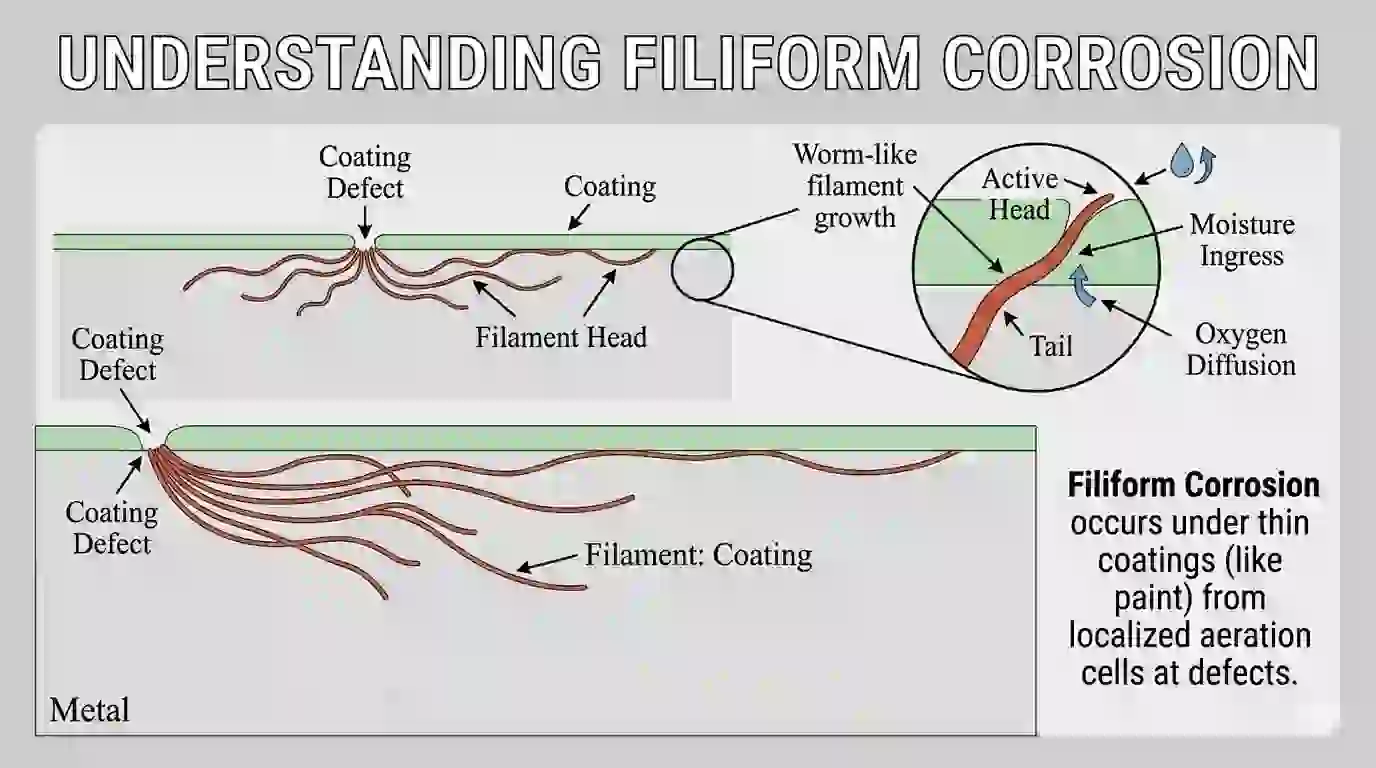
Filiform corrosion happens under coatings like paint.
When moisture enters through a small defect, corrosion starts and spreads under the coating in thread-like patterns. It is common in humid environments.
This corrosion mechanism depends on oxygen and moisture movement under the coating.
You will see thin lines spreading under paint.
Prevention includes proper surface preparation, cleaning, and using high-quality coatings and primers.
7. Stress Corrosion Cracking (SCC Corrosion Type)
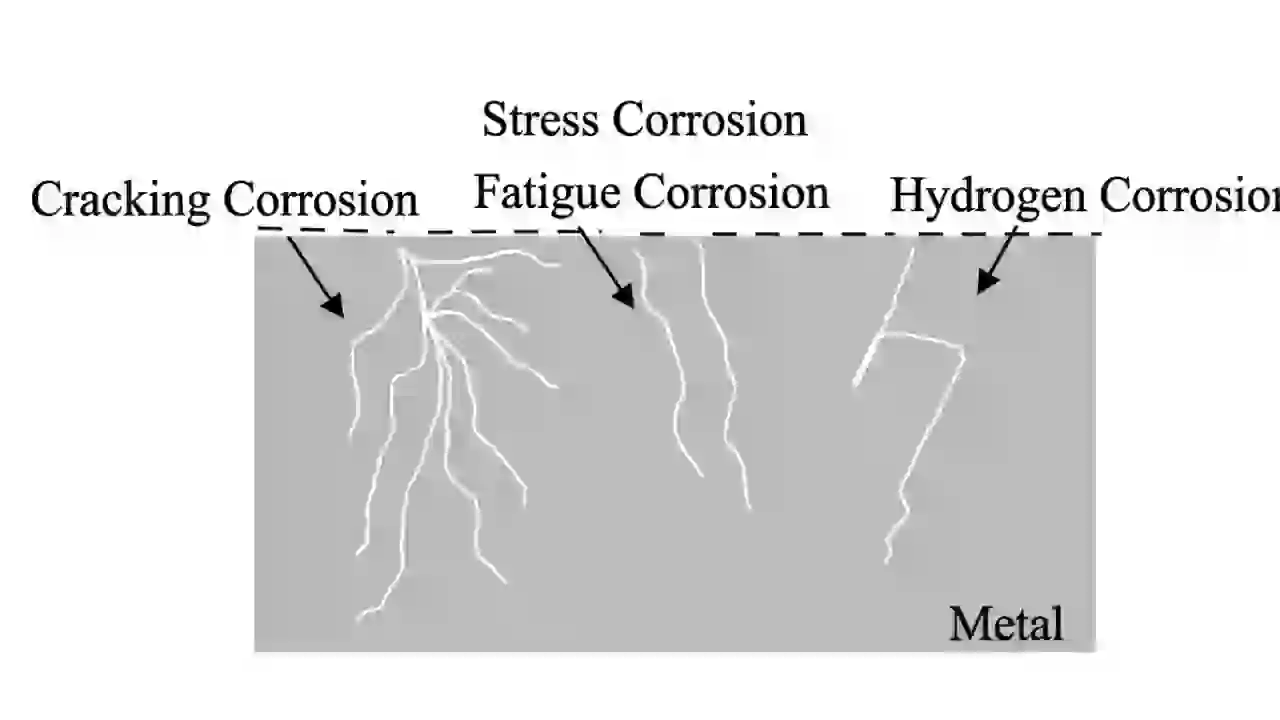
Stress Corrosion Cracking is a serious corrosion type where cracks form due to three factors:
- Tensile stress
- Corrosive environment
- Susceptible material
The cracks grow slowly but can suddenly cause failure without warning.
For example, stainless steel can crack in chloride environments, and carbon steel can crack in caustic solutions.
Prevention includes reducing stress, using proper materials, and controlling the environment.
8. Erosion Corrosion (Flow-Induced Corrosion Type)
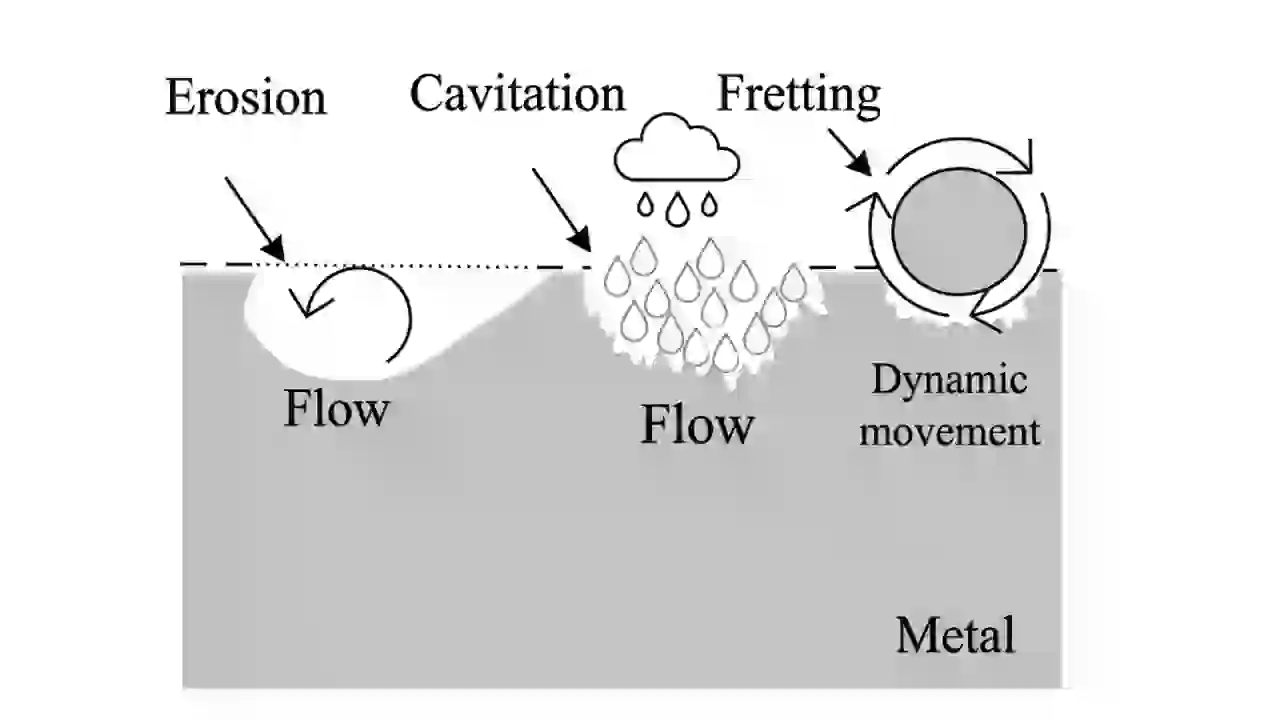
Erosion corrosion happens when fast-moving fluids remove the protective layer of metal.
This exposes fresh metal to corrosion again and again, increasing the corrosion rate.
This type is common in pipelines, pumps, and valves where fluid velocity is high.
You will notice grooves or directional marks on the surface.
To prevent this, engineers reduce flow velocity, improve design (smooth bends), and use erosion-resistant materials.
9. Fretting Corrosion (Wear and Corrosion Type)
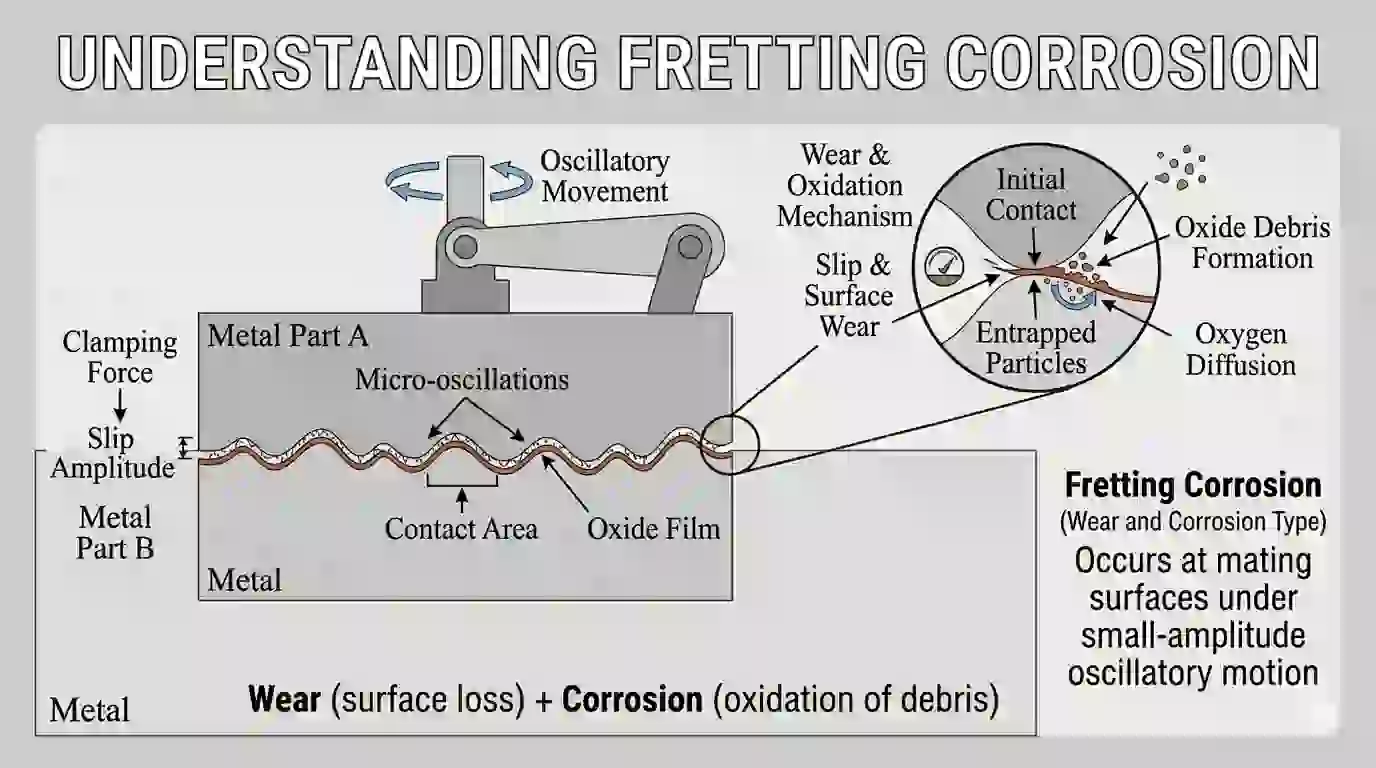
Fretting corrosion happens when two metal surfaces are in contact and experience small vibrations.
These vibrations remove the oxide layer, and fresh metal reacts with oxygen.
This produces fine powder (oxide debris) and causes surface damage.
It is common in bolted joints, bearings, and rotating parts.
Prevention includes lubrication, proper tightening, and surface coatings.
Conclusion: Why Understanding Corrosion Types is Important
Understanding different types of corrosion is very important in engineering and maintenance. Each corrosion type has a different cause, and the prevention method also changes.
If you can identify the corrosion pattern early, you can avoid major failures, reduce maintenance cost, and increase equipment life.
In industries like oil and gas, power plants, and construction, proper knowledge of corrosion types and prevention is essential for safety.
FAQs on Types of Corrosion
What are the main types of corrosion?
The main types of corrosion include uniform corrosion, galvanic corrosion, pitting corrosion, crevice corrosion, intergranular corrosion, stress corrosion cracking, erosion corrosion, and fretting corrosion.
Which corrosion type is most dangerous?
Pitting corrosion and stress corrosion cracking are considered very dangerous because they can cause sudden failure without visible warning.
What causes corrosion in metals?
Corrosion is caused by electrochemical reactions between metal, oxygen, moisture, and chemicals like salts.
Can corrosion be completely stopped?
Corrosion cannot be fully stopped, but it can be controlled using coatings, material selection, and proper design.
Why is corrosion faster near coastal areas?
Coastal areas have high moisture and salt (chloride), which increase corrosion rate, especially for steel.
Suggested Posts
Airless Spray Painting: Simple Guide for Better Coating Quality
3 LPE vs 3LPP Coating System – Complete Guide for Pipeline Coating
Painting Inspection Standards (ISO, ASTM, SSPC, NACE) – Complete Guide for QA/QC Engineers
